Ensuring all shipments records are kept and that drugs are delivered safely
The Coghlan Group is a US company that was founded by experienced clinical supply professionals to provide the global pharmaceutical research industry with a reliable source for the storage, distribution, preparation, packaging/labeling and randomization of clinical trial materials.In 2014, TCG was wrapping up a global Phase 3 clinical study that consisted of approximately 8,000 temperature-monitored shipments when there was a push by the Sponsor to collect all outstanding temperature data from the investigational sites and place it on file in a central location at TCG.
Securing shipment data
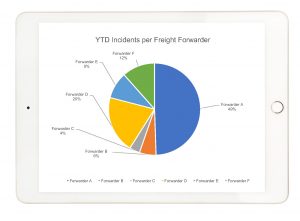
“We recognized that many sites were not following the established procedures and returning the temperature files in a timely manner, if at all. It was a significant effort to retrieve the outstanding temperature records across 400 sites and when we were finished, we barely surpassed 90% of all temperature records collected”, says Mike Butterworth, Clinical Trials Global Program Director at TCG. Adding to the time and effort spent collecting the temperature records was accurately linking them back to their original shipment.This raised a whole host of questions from not only a business process standpoint, but a compliance perspective as well.
How could TCG efficiently confirm that the sites were returning the temperature data to them in a timely manner? For those sites that did not return the data, how could TCG ensure the product was suitable for use? Finally, how could TCG reduce the time spent retrieving the missing temperature records and tracking the files back to their corresponding shipments? “At the time, we were completely reliant upon the sites to be fully compliant to review and send us the data, in addition to alerting us in a timely manner when shipments alarmed”, Butterworth says.
The solution
Cloud temperature management solution .To reduce the time spent finding the temperature records and tracking it to the right shipment, TCG wanted a solution that could automatically link the temperature data to each kit without having to allocate significant staff resources to manually type in this information. “We wanted to increase compliance and visibility, but it had to make sense from a business perspective”, says Butterworth. TCG worked with TSS and their IWRS provider to develop a barcode solution that is now printed on each shipment request.
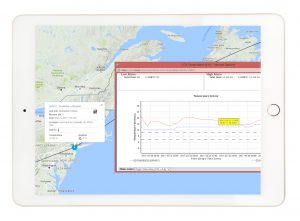
The barcode is unique to each shipment and contains information that ties the temperature data back to a specific drug unit or kit number. “With just one barcode scan, the data is instantly loaded into the TSS cloud temperature management solution and the TempTracer data logger is automatically launched”, Butterworth explains. To make the process even more effective, TCG has incorporated a label printer that automatically produces a label with shipment-specific information at the time of scanning.
The result
Efficient and compliant shipments According to Butterworth, using the TSS solution has resulted in great improvements in operational efficiencies at TCG, as well as increased visibility across the different clinical sites. “The system allows our depots and the CRAs to monitor site compliance remotely and in real-time. When an upload has not entered the system after a certain period of time, we follow-up to ensure we collect all data before they potentially discard the TempTracer”, says Butterworth. This means TCG no longer has to spend lot of time hunting the data, as all shipments can be accessed in the cloud. “The total response time from receipt-to-review of an alarmed shipment has been significantly reduced and can be tracked for a quality metric”.
Furthermore, the cloud solution has allowed TCG to effectively evaluate the risk-based approach they have recently implemented in a large global study in regards to transit temperature controls. ”Temperature trends, transit times, alarm rates and compliance can all be evaluated quickly and easily in the system”, Butterworth concludes.

The World’s Leading Temperature Management Solution